Adapted from the Journal of the Heartland Angels
Adapted from the Journal of the Heartland Angels
By John Jonelis
ADMdx has pioneered a new science to help big pharma wipe out Alzheimer’s disease. With a mission that huge, they can pivot and adjust, yet never stray from their course. I’m talking to their CEO, Dawn Matthews and her first statement knocks my socks off.
.
The Urgent Need
Dawn Matthews—Up until now, all of the Alzheimer’s trials have unfortunately failed. It’s a needy market.

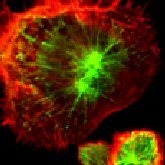
That jolts me and I shove my coffee aside. Dawn fires up her computer and shows me a series of images—brains lit up with bright red and blue blotches. I look at the scans of diseased brains and wince.
.
Dawn—When a patient expresses a pattern, it’s important to know what it would look like over time. Here are people with Mild Cognitive Impairment. Then you see deficits emerge. As it worsens, this pattern becomes more and more pervasive.
John—Not pretty pictures.
Dawn—Not at all. Alzheimer’s is a disease that progresses over maybe twenty years. In this disease, the neurons stop functioning and die. By the time doctors see symptoms, these changes are pretty advanced.
D—There is thinking in the medical community that the earlier we can intervene, the better.
I take that as a yes.
..
The Solution
D—The good news is we’ve discovered that Alzheimer’s has a very characteristic fingerprint:
- We can now detect it LONG BEFORE symptoms appear.
- We can tell whether it’s Alzheimer’s or another form of dementia.
- We can predict the rate at which someone is likely to decline.
J—That sounds a whole lot more hopeful. There must be a huge demand for that.
D—There is. We conduct imaging studies for pharmaceutical companies and we help them understand whether their drug is working. Say a drug company collected data for a big clinical trial. They want to know answers such as:
- Is the drug generally effective?
- What are the responses at different doses?
- Is the drug viable to bring into a larger clinical trial?
D—Larger trials are expensive, so—
J—So they can target what they do. That sounds incredibly valuable.
D—It is. One company actually used our data to kill a drug. They saw VERY EARLY that the required dose would cause unacceptable side effects.
.
Brain Scan 101
D—This is a CT scan—it measures tissue. This one’s an MRI—it shows brain anatomy in exquisite detail. Here’s a PET scan—it measures chemical activity. Here, we’ve superimposed PET and MRI imagery to give an anatomy reference to our analysis. The colors you see are from a radioactive tracer.
For Alzheimer’s research, we use a tracer that binds to amyloids (inappropriate proteins). Now, in Alzheimer’s disease, you see a lot of amyloid plaque. The diagnostic criterion at autopsy is the presence of a certain number of plaques and a certain number of tangles.
That phrase, “at autopsy” is a bit chilling for me, but I stuff my feelings and ask the next question.
J—It’s something like hardening of the arteries?
D—It is.
J—So you find amyloid plaque, using PET. You fit that to an MRI showing brain structure and detect Alzheimer’s before anybody knows it’s there.
D—Yes!
Reaching for my coffee again, I feel a grin coming on. I just aced a quiz!
.
Reading the Mind
J—What’s actually unique here?
D—Pattern recognition and automation. Different kinds of dementia have overlapping symptoms and it’s impossible to differentiate by conventional means. It’s not always possible to tell what’s going on by just eyeballing a scan. That’s why an automated algorithm is crucial. With machine learning, we can read the fingerprint of the disease. Over time, the pattern becomes more and more pervasive but each keeps its unique fingerprint.
J—Sounds like chart patterns in algorithmic futures trading.
D—Yes, Yes! Perfect analogy! We combine subjects and perform group comparisons:
- We compare low and high doses to the placebo.
- We see if the drug is binding to a certain receptor in the brain.
- We determine patient suitability. That’s important to run a valid study.
.
I get a strong sense that Dawn Matthews is passionate about her work.
.
The Secret Sauce
D—Scans are costly. If you want to point to one technical differentiator, it’s the methods we use to extract data from images generated from almost any scan available. Our secret sauce is the way we analyze them.
J— You don’t perform scans yourself?
D— We don’t. The data is electronic and it comes to us. We process and analyze it centrally. It’s true that a lot can go wrong when taking a scan, but it’s better to be proactive than limit your database to only the highest quality scans. We have been fortunate in that. We have over 5000 scans in our repertoire and feel we’ve reached critical mass.
J—That data must be noisy.
D—It is. Because clinics perform the scans, there is different scanner hardware noise, different detector noise, different doses of tracer, and differences in how the tracer is absorbed. The raw signal changes from scan to scan and from person to person.
J—So signal-to-noise ratio is an issue. What does the medical community do about it?
D—Currently, they don’t do anything. What you get is something really distressing. Big companies can’t make sense of their data. Even at the best universities, known for their Alzheimer’s programs, there’s about a 20% rate of misdiagnosis.
.
What she’s telling me is that conventional methods find the disease way too late and even then the chance of a correct diagnosis is worse than Russian Roulette.

D—But we’ve solved the noise issue. Our pharma partners tell us what we’ve done is a game changer. We’ve built two very sophisticated platforms. We can innovate rapidly. We can perform drug studies and automatically create an audit trail that records everything that happened to the data. It’s 21CFR Part 11 validated and can do large Phase III clinical trials.
J—That’s all in place?
D—Yes.
J—It’s artificial intelligence?
D—It’s machine learning. The program finds the COMBINATION of patterns (because it’s not just one pattern) that best discriminate these different classes. During accuracy tests, we scored 100%. I’m not saying that we’ll always be exactly right, but it’s highly accurate.
J—You don’t have to go through Clinical Trials—Phase I, Phase II, Phase III?
D—No, no—that would be for a drug company. We already did the work to get compliant for the studies we do. Going forward, there’s no reason we can’t successfully grow our business.
J—Any buyout prospects?
D—Yes, we’ve already been approached. Our expectation is that a large company will partner with us.
J—Well well – It sounds to me like, “Goodbye Dr. Alzheimer.”
And when ADMdx cracks that one, they can bring their platform to bear on the next big problem.
.
This article is adapted from the Journal of the Heartland Angels
Download NEWS FROM HEARTLAND (1.2 MB PDF)
.
Contacts
ADMdx: http://admdx.com/
Email: inquiries@admdx.com
Phone 847-707-0370
Photo credits – ADMdx, Wikipedia
.
NEWS FROM HEARTLAND, The Journal of the Heartland Angels, is a quarterly newsletter published as an information service to its members. Articles may be reproduced with attribution for educational purposes. Copyright © 2013 Heartland Angels – John Jonelis, Editor
CAVEAT EMPTOR: This article is for educational purposes and is not investment advice. All investment involves substantial risk. Please do your due own diligence.
Contact Ron Kirschner – Ron@HeartlandAngels.com
For more information, go to: www.HeartlandAngels .com
.
Chicago Venture Magazine is a publication of Nathaniel Press www.ChicagoVentureMagazine.com Comments and re-posts in full or in part are welcomed and encouraged if accompanied by attribution and a web link . This is not investment advice. We do not guarantee accuracy. It’s not our fault if you lose money.
.Copyright © 2014 John Jonelis – All Rights Reserved
.
.