 Essential Biotechnology –
Essential Biotechnology –
New Treatment for Hard to Treat Cancer
Jayson Kurfis
A protein, CRR9, serves as a nexus for tumor cell survival. It’s located on the cell surface, making it an accessible drug target. By developing antibodies to the CRR9 protein, Essential Biotechnology has devised a method to kill therapy-resistant tumors.
The Problem
While researchers have made significant strides in cancer treatment, patient outcomes vary widely based on type. As Table 1 shows, lung and pancreatic cancer have a very high mortality while skin cancer has a five year survival rate of over 90%. In the case of ovarian cancer, the initial tumor is treatable; however, secondary tumors often resist treatment. The secondary tumors almost always result in the patient’s death.
Resistant ovarian tumors are typically associated with later diagnosis. And, unfortunately, late diagnosis is common because ovarian cancer often has mild symptoms that are either ignored or attributed to other causes. Due to these facts, early diagnosis of the disease only happens 20% of the time. Hence, a majority of patients develop secondary cancer that resists existing therapies. At this point, there is no standardized treatment when this occurs, leading to significantly higher mortality.
The Target
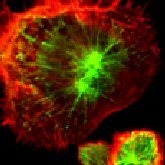
In 2001, researchers discovered a protein called CRR9 that is upregulated in chemotherapy resistant ovarian cancer cells (4, 12). Further investigation demonstrated that this protein serves as a nexus in regulating tumor cell survival (Figure 1) and is located on the cell surface making it an accessible drug target (16).
While initial studies showed that CRR9 was associated with treatment resistance in ovarian cancer, additional studies were completed to see if other cancers utilized the same protein to resist treatment. It was later found that the CRR9 protein was associated with lung and pancreatic cancers (5). These two forms of cancer have among the worst 5 year survival rates of any cancer and expression of this protein was associated with poor patient outcomes (8, 10). Figure 2 demonstrates the success of using a CRR9 inhibitor against lung cancer in an animal model (16, Figure 2).
Essential Biotechnology:
The Proposed Medication
By developing monoclonal anti-CRR9 antibodies to specific parts of the CRR9 protein, Essential Biotechnology devised a method to kill therapy-resistant and metastasizing tumors by impacting multiple mechanisms of tumor cell survival. Further, due to the specificity of the target and the antibody being humanized, there is a reduced likelihood of side effects. This specificity also leads to a more effective therapy that has a better chance of killing the cancer cell. Preclinical work has already shown that this approach is highly effective, even on traditionally “undruggable” Ras-driven cancers, the most common example being lung (Figure 2).
Essential Biotechnology offers an integrated product pipeline that targets a novel mechanism of tumor survival, growth, and progression that is highly effective and synergistic with existing therapies, such as genotoxic chemotherapy. Sensitization to existing drugs will reduce the necessary dose and toxicity leading to decreased morbidity, metastasis, and toxicity. Ultimately, this results in decreased medical costs and increased quality of life for the patient.
.
Competitive
While there are molecular pathway inhibitors for other types of cancer that are currently at the clinical trial phase, these are downstream from CRR9 (Figure 1). This means that competitive drugs inhibit one arm of a molecular mechanism that actually utilizes multiple pathways (Figure 1). CRR9 impacts far more than one leading to an improved outcome and decreased chance of acquired resistance to anti-CRR9.
.
Technology Protection and Competitive
At this point, the work is being completed at the Medical College of Wisconsin and a US patent has been issued (Table 2). In addition, an application was submitted to cover the technology across most other countries (Table 2). The intellectual property is managed by the Medical College of Wisconsin’s Office of Technology Development.
Goal
To realistically balance the short-term potential with the development cost for the therapeutics, the leadership of Essential has decided to focus on ovarian cancer. While this disease impacts less than 200,000 patients per year, it does represent an unmet clinical need that could be very profitable (Table 1). Due to the market size and the FDAs interest in fast-tracking drugs for resistant ovarian cancer, any drug that addresses this need is eligible for orphan status and has a high likelihood of being fast-tracked through the FDA. Further, there could be tax advantages for development due to the high level of mortality associated with this disease and the importance in finding a suitable treatment. Additionally, the company is pursuing non-dilutive seed funding through the federal small business technology transfer, STTR, program, which, if received, will abrogate the cost of development while not diluting equity stakes taken by angel or other investors.
Essential Biotech is working towards completion of pre-clinical studies that will enable the company to submit an investigational new drug application with the FDA. Upon completion of these objectives, Essential will start phase I trials. Due to the clinical need, the FDA is likely to fast-track development and may also provide tax incentives for development. At the conclusion of phase I, Essential will look to exit and partner with a pharmaceutical company that has the resources to move the drug through the additional phases of clinical development.
.
Profit/Planning
Essential has analyzed current and future spending estimates and evaluated the penetration of comparable targeted biologic therapeutics, which are driving a higher rate of market growth compared to other cancer therapeutics. To estimate rate of return, the company thoroughly investigated comparable deals specific to biologics in oncology and conducted risk-adjusted net present value analysis with industry aligned professionals. Additionally, Essential used information from the Licensing Executives Society, Thompson Reuters professional analyses and meta-analyses of published studies by patient advocacy organizations. Royalty revenues in the initial market could reach $1 bn for Essential’s biologic over the life of the patent. If applied to the broader market, it is possible to reach the 1 billion dollar amount annually. Because developing treatments in this area is an unmet clinical need, the market appetite indicates that any proof of concept through phase I will generate significant interest from pharmaceutical partners.
Essential Biotechnology is further seeking investment for product optimization and progression of therapeutics including monoclonal antibody EB 10-2 through Phase I/II clinical trials. We anticipate completion of Phase I trials and proof of concept in humans, with licensing and upfront ROI by the end of 2018.
.
Corporate Leadership Position
Essential’s CEO has over 20 years of experience in biotech leadership. In addition, the CSO has preeminent scientific expertise with the CRR9 target as is evidenced by the numerous articles and patents that he has written on this topic (4, 5, 13-15). Further, because of the companies association with the Medical College of Wisconsin, the company has access to numerous scientific advisors with extensive experience in clinical oncology as well as a clinical trials director associated with the hospital.
Thus far, the company has won $50,000 from the Health Innovation Pitch event that took place in December of 2015. This event brought together local, national and international angel investors as well as industry professionals from Novartis and OrbiMed (18). Essential Biotechnology is among six finalists for the LES Foundation International Business Plan Competition and in the final competition for the Wisconsin Governor’s Business plan contest.
■
Potential investors: Please contact us for information: invest@essentialbiotech.com\
Check them out on LinkedIn: https://www.linkedin.com/company/essential-biotechnology
.
References
- Puskas LG, Man I, Szebeni G, Tiszlavicz L, Tsai S, James MA “Novel Anti- CRR9/CLPTM1L Antibodies with Antitumorigenic Activity Inhibit Cell Surface Accumulation, PI3K Interaction, and Survival Signaling” Mol Cancer Therapy Mar 3, 2016 –epub
- James MA, Vikis HG, Tate E, Rymaszewski AL, You M. “CRR9/CLPTM1L Regulates Cell Survival Signaling and is Required for Ras Transformation and Lung Tumorigenesis” Cancer Research 74(4) Dec. 2013
- Yamamato K, Okamoto A, Isonishi S, Ochiai K, Ohtake Y “A novel gene, CRR9, which was up-regulated in CDDP-resistant ovarian tumor cell line, was associated with apoptosis” Biochem Biophys Research Communications. 2001 Feb 2; 280(4): 1148-54
- Michael A. James,1 Weidong Wen,2 Yian Wang,2 Lauren A. Byers,3 John V. Heymach,3 Kevin R. Coombes,3 Luc Girard,4 John Minna,4 and Ming You1 “Functional Characterization of CLPTM1L as a Lung Cancer Risk Candidate Gene in the 5p15.33 Locus” PLoS One. 2012;7(6):e36116
- James MA, Seibel WL, Kupert E, Hu XX, Potharla VY, Anderson MW. “A novel, soluble compound, C25, sensitizes to TRAIL-induced apoptosis through upregulation of DR5 expression”
- Essential Biotechnology website (http://essentialbiotech.com/)
- Kathleen Gallagher “Essential Biotechnology LLC wins top prize for health pitch event” Dec. 7, 2015. Milwaukee Journal Sentinel. http://www.jsonline.com/business/essential-biotechnology-llc-wins-top-prize-for-health-pitch-event-b99629932z1-360851931.html
- Jeff Engel “Bridge to Cures Eyes More Funds, Bigger Programs to Nurture Startups” Dec. 24, 2015 Xconomy Wisconsin. http://www.xconomy.com/wisconsin/2015/12/24/bridge-to-cures-eyes-more-funds-bigger-programs-to-nurture-startups/
Image Credits – Essential Biotechnology, MS Office
This article first appeared in NEWS FROM HEARTLAND
Chicago Venture Magazine is a publication of Nathaniel Press www.ChicagoVentureMagazine.com Comments and re-posts in full or in part are welcomed and encouraged if accompanied by attribution and a web link. This is not investment advice. We do not guarantee accuracy. It’s not our fault if you lose money.
.Copyright © 2016 John Jonelis – All Rights Reserved
.
.